Using HBI in Blast Furnaces

The Paris Agreement, with the objective of a maximum global increase in temperature of 2°C by the end of the century compared to the pre-industrial period, is undoubtedly an important step toward true global climate protection. The European Green Deal is a response to these challenges and defines strategies to transform the Union into a modern, resource-efficient, and competitive economy. The overarching goal of the Green Deal is to make Europe the first GHG-emission neutral continent by 2050.
TOWARD CO2 EMISSION-FREE STEELMAKING
Where do the challenges lie in the decarbonization of steelmaking? CO2 emissions originate from the coke/coal-based blast furnace (BF) and basic oxygen furnace (BOF) process route, which—still today—is the global standard for 72% of worldwide steel production (see Figure 1). Carbon is needed in the production of hot metal in the BF as a reducing agent for iron oxides and supplies the process energy required for gasification. The carbon contained in the hot metal must be oxidized through top blowing in the BOF converter. Carbon and oxygen together form carbon dioxide. A reduction of process-related CO2 emissions can only be achieved by partially (or even completely in the long term) replacing carbon with hydrogen.
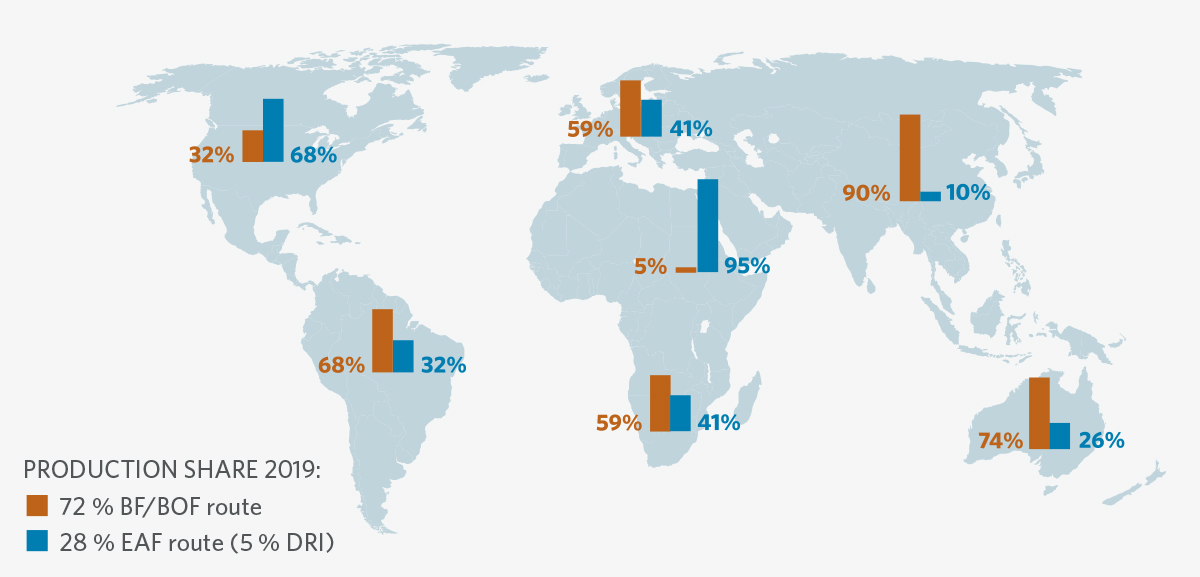
FIGURE 1.
Global steel production routes [www.bir.org (11th Edition World Steel Recycling)]
Gradual Decarbonization
From today’s perspective, the scenario to achieve the climate targets includes a gradual decarbonization of steelmaking with a long-term vision of completely replacing CO2 with hydrogen. The interim technology relies on the direct reduction (DR) process where natural gas is used to produce direct reduced iron (DRI) in the densified form of hot briquetted iron (HBI). With respect to the core process of DR, this results in a CO2 reduction of 50% when compared to the BF process. Depending on the generation capacity for power used to melt scrap in the electric arc furnace (EAF), a reduction of around 35% can be achieved by the DR/EAF process when compared with BF/BOF route. It is thought that CO2 emissions could be further reduced to 20% of the BF/BOF route when Proton Exchange Membrane (PEM) electrolyzers are used with the DRI/EAF route (see Figure 2).
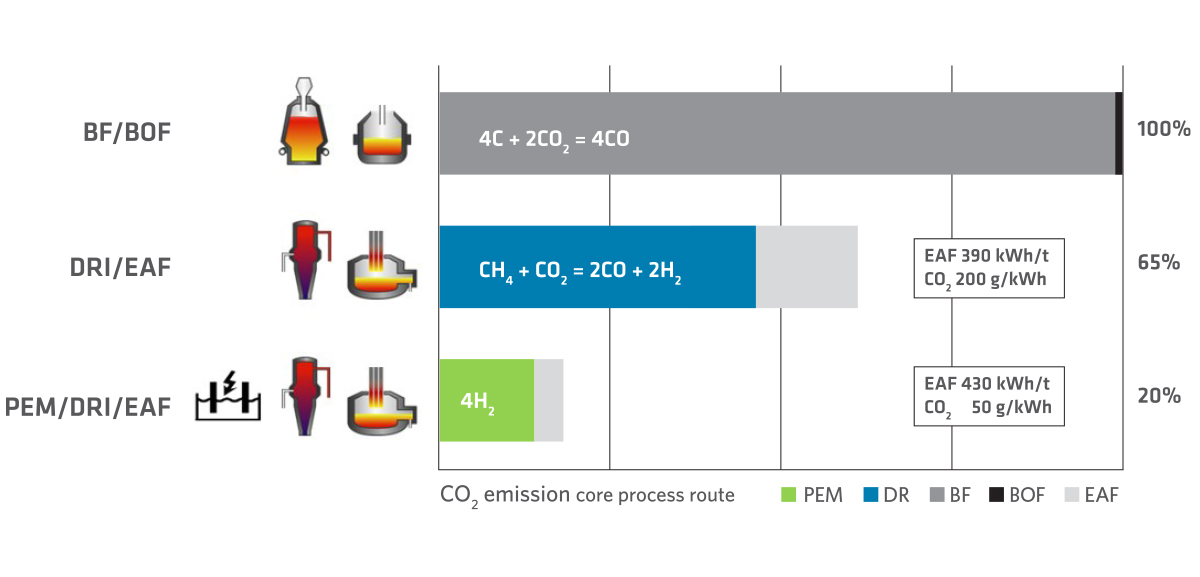
FIGURE 2.
CO2 emissions of steel production routes
HBI for BF Operation
The potential for HBI to be used in the BF/BOF steelmaking route rather than just EAF-based steelmaking, with which the use of all forms of DRI are usually associated, could play a significant role in the future of European iron and steelmaking with the target of CO2 reduction. HBI has been used successfully by steelmakers in the USA for 25 years, so there is no technology risk associated with using it compared with breakthrough technologies. voestalpine in Linz, Austria uses HBI from its own plant in Corpus Christi, Texas, USA, (Figure 3) in its blast furnaces. This article will discuss the influence of the usage of HBI in the blast furnaces on the consumption of reducing agents, productivity, blast furnace operation, and hot metal quality.

FIGURE 3.
voestalpine Texas HBI plant
Blast Furnaces 5 and 6 at voestalpine Linz have a hearth diameter of eight meters and on-average produce 2500–2700 tons of hot metal per day. The specific quantity of HBI used in the blast furnaces for a selected period and the total cumulative consumption are shown in Figure 4. The HBI quantity peaks were at 160kg/t HM. The briquettes are charged with the burden in the blast furnace.
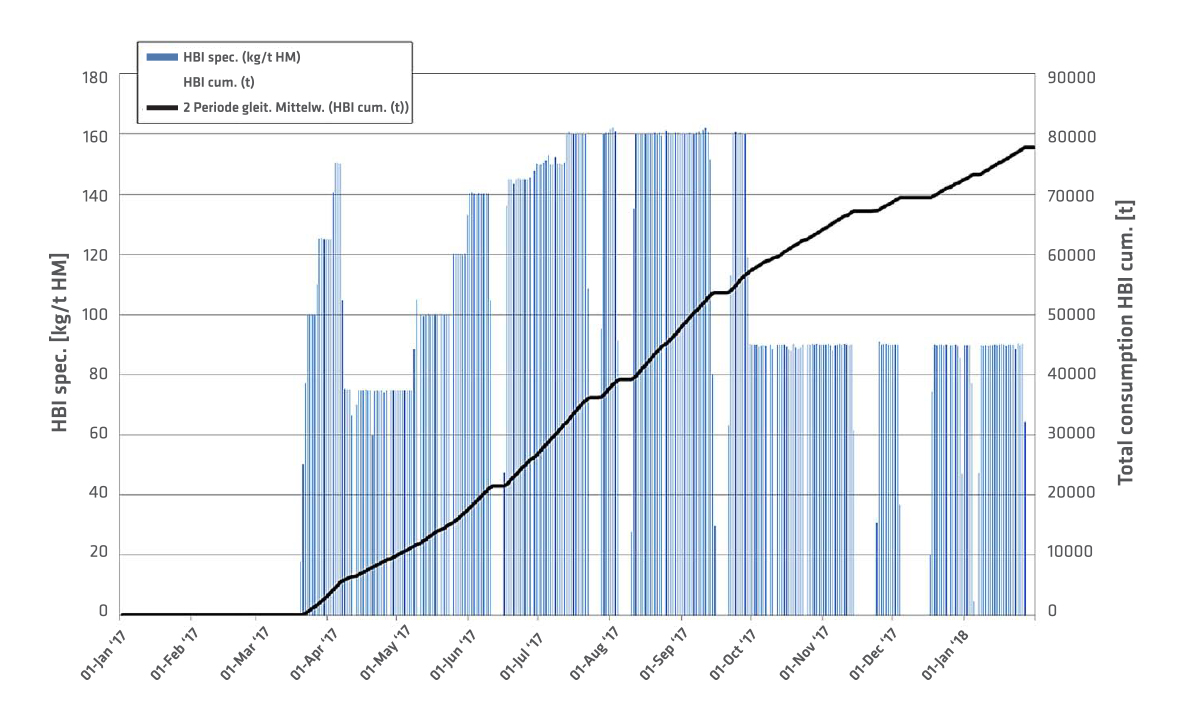
FIGURE 4.
HBI quantity used in BF operation
Influence of HBI on Reducing Agents and Melting Rate
Typically, HBI contains over 90% Fe and the metallization degree is higher than 90%. These metallics only need to be melted and hardly reduced; therefore, HBI in BF operation decreases the consumption of reducing agents. In our experience, if 100kg HBI/t HM is used, the reducing agent rate (coke equivalent) can be decreased by approx. 25kg/t HM, as shown in Figure 5. The colors indicate the amount of oxygen in the hot blast, and the amount of oxygen lies between 4000–6000Nm3/h for more than 60% of the data points. If the coke rate is reduced, the rate of PCI (pulverized coal injection) has to be adapted. Coke can never be eliminated (although HBI is used) because coke has to provide the structure and the permeability in the shaft. Therefore, a minimum coke rate has to be guaranteed. The productivity (melted iron [t] per hour) can be increased if HBI is charged in the BF. Productivity can be raised up to 10% per 100kg HBI/t HM (Figure 6) at constant oxygen level.
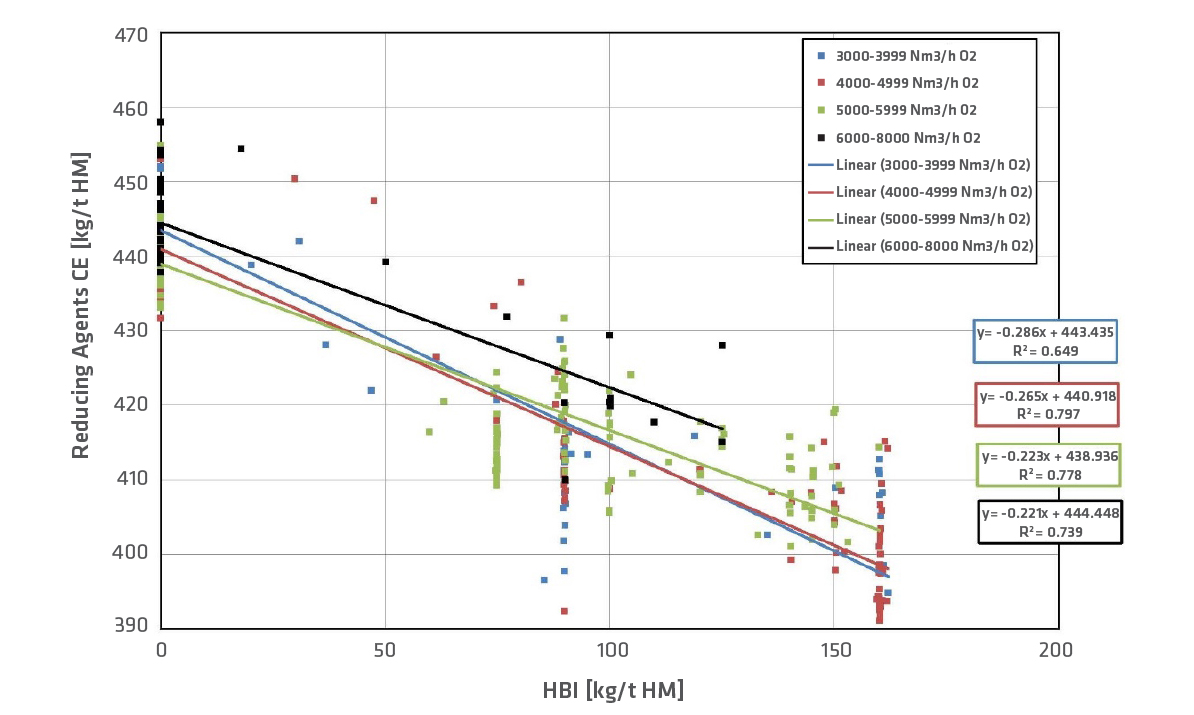
FIGURE 5.
Development of reducing agents with HBI
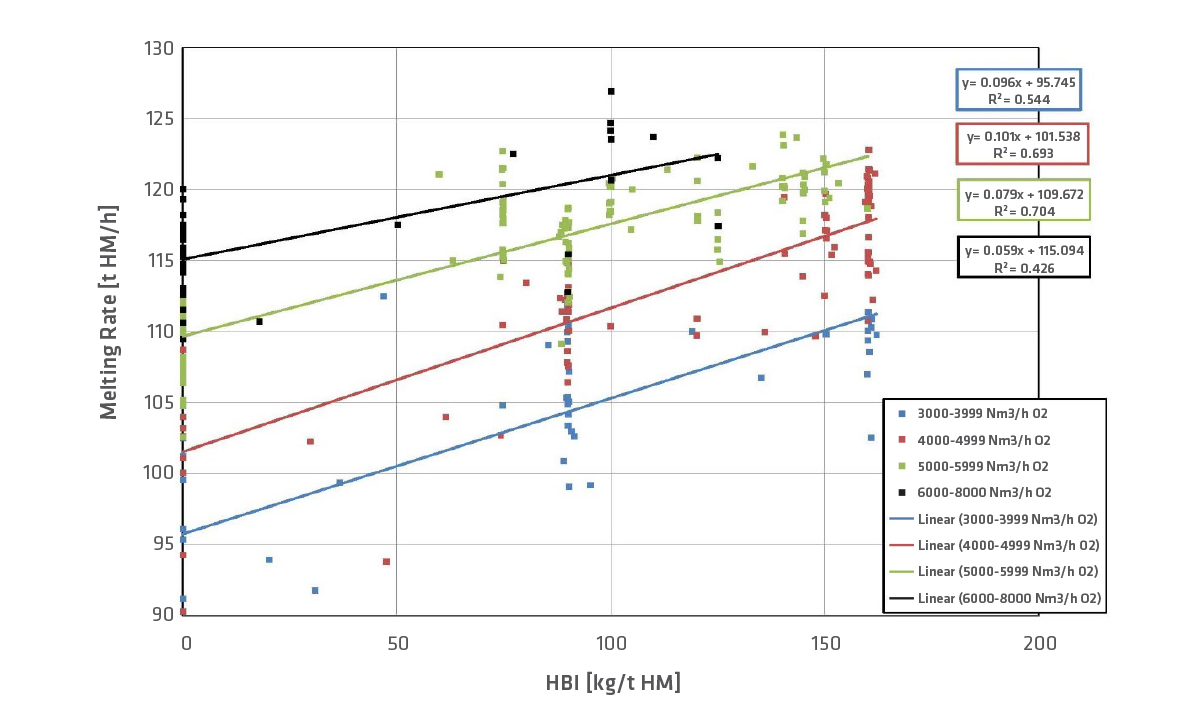
FIGURE 6.
Development of melting rate with HBI
Influence of HBI on Top Gas Conditions
The gas utilization decreases between 0.5-1% per 100kg HBI/t HM (Figure 7). So, the percentage of CO2/H2O in the gas decreases, while the percentage of CO/H2 rises. HBI is a pre-reduced material; therefore, there is less iron oxide to be reduced in the BF. Because the percentage of CO and H2 in the gas is higher with the use of HBI, the calorific value increases slightly, up to 1.5%.
No significant correlation was found for the top gas temperature. During the different test periods, there was no influence of HBI on the cooling capacity and no significant gas flow was on the BF walls.
Permeability of the shaft has not shown any negative or positive reaction when HBI was in the burden. This is also a sign that HBI does not trigger gas flows along the walls in the furnace or influence the furnace operation in a negative or positive way.
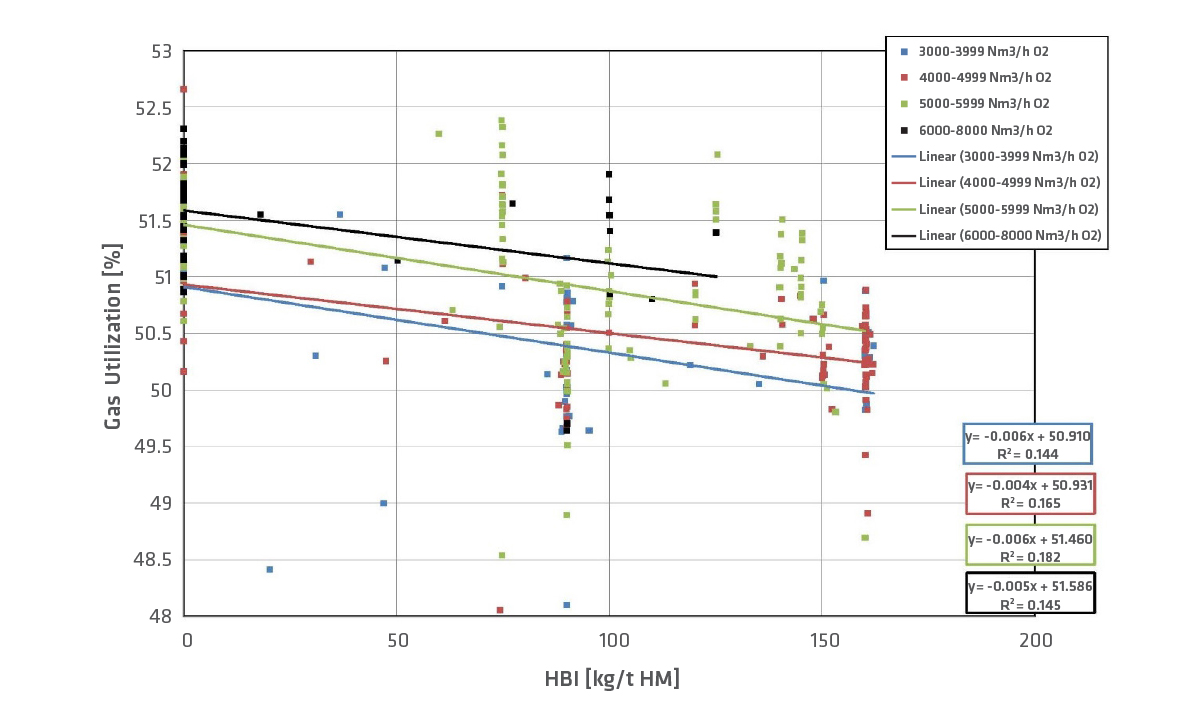
FIGURE 7.
Development of gas utilization with HBI
Influence on HBI on Hot Metal Quality
An indication of high hot metal quality is high carbon content and low sulfur content. Reducing agents have high sulfur content (coke has approximately 0.5–0.7%) and the sulfur blocks the carburization of hot metal. The coke rate can be decreased if HBI is charged in the BF. Figure 8 shows the S-content of hot metal at an HBI charge of 100–150kg/t HM. A lower S-content of hot metal is reached with an HBI charge.
But the S-content of hot metal does not depend only on the rate of reducing agents but also on slag rate, use of recycling material with high amounts of sulfur, and the melting rate (at higher melting rates, the coke stays in the hearth short-time).
A higher C-content of hot metal is achieved with an HBI charge of 100–150kg/t HM (Figure 9). Sulfur retards the carbon solution in hot metal; therefore, there is a higher carbon content in HM (=less sulfur input in case of HBI).
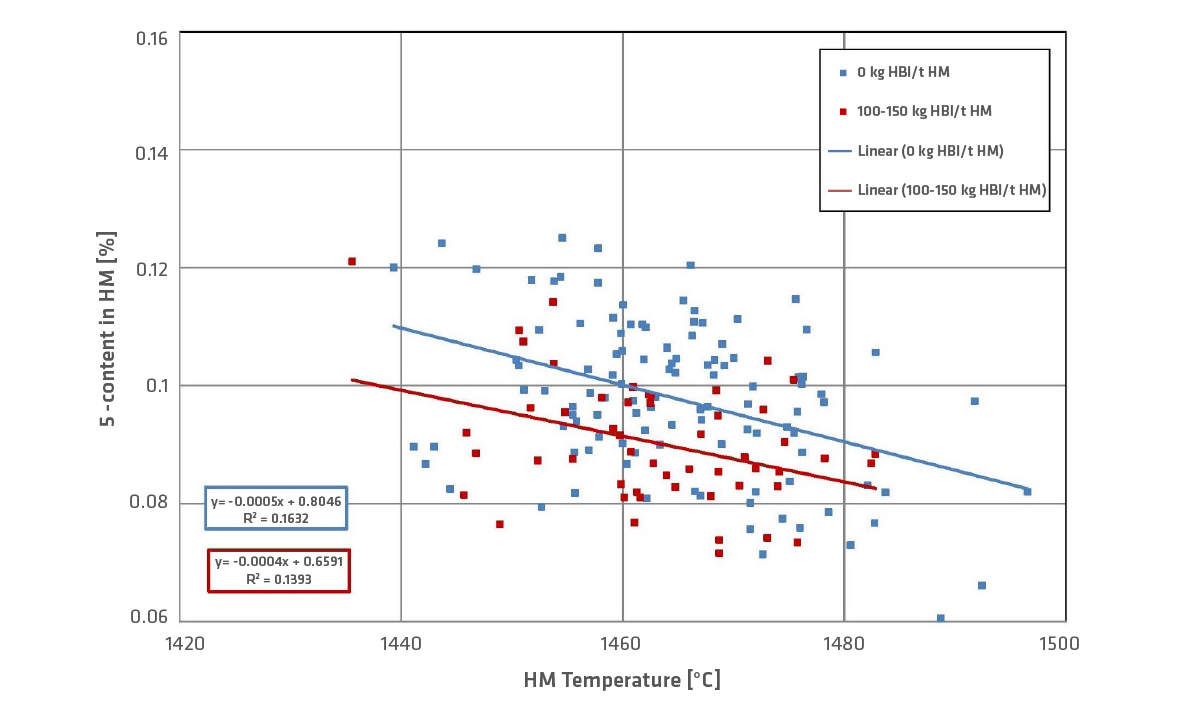
FIGURE 8.
Sulfur content in hot metal
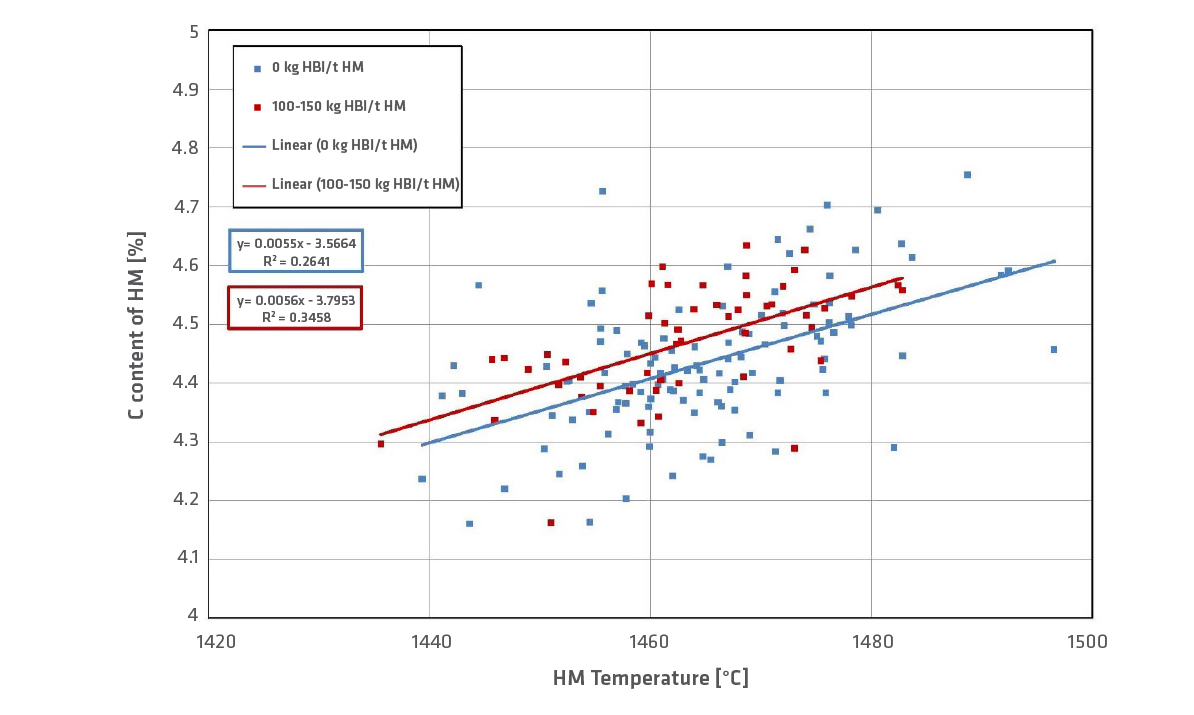
FIGURE 9.
Carbon content in hot metal
Conclusions
The use of different charging rates of HBI from the voestalpine Texas plant in Corpus Christi, Texas, in BFs at voestalpine Linz was examined over the period from 2017-01-01 to 2018-01-31 based on daily average data. When 100 kg HBI/t HM is charged to the BF, the following conclusions can be drawn:
- Reducing agents (CE) can be decreased by 21.9 – 27.5 kg/t HM, whereas the coke rate can be decreased by 10.9 – 18.1 kg/t HM.
- Productivity can be increased up to 7.3 – 10.1% at constant oxygen levels.
- Gas utilization drops by approximately 0.4 – 1.1% because HBI is a pre-reduced material and less oxide is charged to the BF. The decrease in quantity of charged oxide also reduces the quantity of
CO and H2 which are transformed into CO2 and H2O. - The calorific value of top gas rises by 12.3 – 23.5 Wh/Nm3 due to the higher contents of CO and H2.
- A correlation between the top gas temperature, cooling capacity, and permeability and the use of HBI cannot be seen.
- The S-content of hot metal is lower and C-content higher (high quality) with a charge of 100 – 150 kg HBI/t HM compared to when no HBI is charged. The S- and C-content in hot metal does not depend only on the charged HBI quantity, which reduces the sulfur containing reducing agents, but also on other factors, such as the slag rate, the quantity and nature of the high S-content recycling material, and the melting rate.



