The Value of DRI – Using the Product for Optimum Steelmaking

Using the Product for Optimum Steelmaking
INTRODUCTION
The use of direct reduced iron (DRI) and DRI products is constantly on the rise and will be for the foreseeable future. DRI production in 2013 hit 75.22 million tons compared to only 23.65 million tons twenty years prior in 1993 and only 49.5 million tons in 2003. Accordingly there are very few individuals even remotely associated with the iron and steel industry that do not know of DRI and its forms as Hot DRI (HDRI), Cold DRI (CDRI) and Hot Briquetted Iron (HBI). But DRI like other metallics can vary in its makeup. There are numerous product properties but only a few that have an immediate, direct and significant effect on the overall iron/steel production process, especially in the electric arc furnace (EAF). Optimizing these variables relative to one another is critical to reducing the total operating cost of iron and steel production. The question often asked is, “What are the properties of the best DRI?” Although some would have us to believe there is one utopian answer, the truth is remarkably different. The better question to ask may be “how can DRI best help our operations.” At AM Montreal, our objective is to maximize liquid steel output while reducing the utility costs to an optimum level. The past four decades of operations has led us to determine that metallic content and physical quality are paramount.
SITUATION
To set the stage, ArcelorMittal not only has the largest capacity for DRI production in the world, the company also has the complete suite of DRI process technologies, operating both MIDREX® Process and HyL Process shaft furnace/reactor plants as well as rotary kiln plants. Further, our history of DRI production is extensive. Our MIDREX® Module 1 at Arcelor Mittal Montreal has been in operation since 1973. The reason we operate these direct reduction plants is very simple; we need high quality low residual metallics for production of steel via the most cost effective route possible.
The use of DRI for ArcelorMittal in general, and more specifically at ArcelorMittal Montreal, is dependent on several factors including; costs versus that of comparable scrap, utility price of natural gas and electrical power, specification for the steel being produced and environmental impact. As with any issue, there are both positive and negative aspects of using DRI. Figure 1 indicates the relative movement of the price for DRI versus Busheling. One can easily see that there is in most cases a raw cost advantage with charging DRI versus scrap and at this time the total operating cost of steel production with DRI is $50 per ton less than that using Busheling.
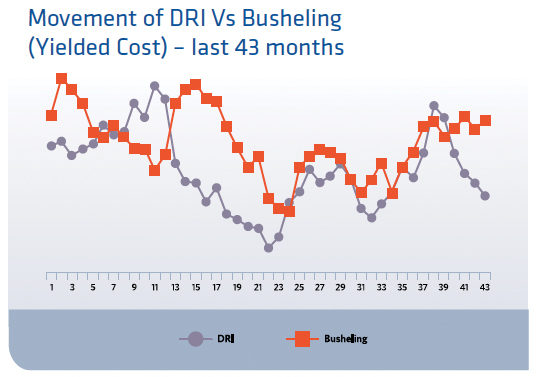
FIGURE 1.
Relative Pricing Movement DRI vs. Busheling May 2012 - November 2014
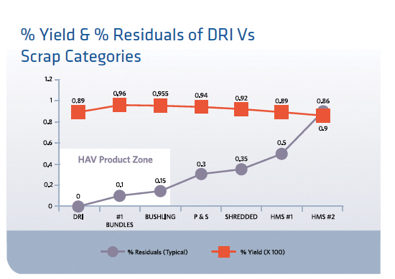
FIGURE 2.
EAF Yield and Residuals versus Charge Material
USING DRI
The advantages of DRI include: predictable chemistry, low tramp material content, carbon and foamy slag formation, lower capital and operating costs, relatively uninterrupted continuous iron making, EAF feed adjustment via blending with lower quality feed material (usually scrap), and less back charging. Figure 2 indicates EAF yield variation when charging DRI compared to scrap loading. It is quite obvious that charging DRI is advantageous to hot metal quality.
Notwithstanding these advantages there are disadvantages as well. These include: additional energy to melt gangue material, increased slag handling, potential for DRI chute blockage, increased baghouse dust and fines, higher refractory costs, lower yield and, and in the case of CDRI versus HBI, there are additional procedures that must be taken for storage, handling and transportation.
With all of this being said, the advantages of DRI in regards to its value to steelmaking operations far outweigh the disadvantages. Being in a position of supplying DRI primarily for internal use we frequently monitor and test the quality of our DRI to assure we are meeting the needs and expectations of our “customer” which in this case are our own meltshops.
Of course DRI is not 100% Fe. It inherently contains some portion of non-reduced iron oxide, carbon and any other components contained within the feed material. Whatever the case, the primary reason for feeding DRI is its iron content. Normally DRI produced from an “EAF grade” iron oxide material, formally referred to as “DRI grade”, will contain 91% to 93% total iron depending on the initial feed material properties. Direct reduction, as the name implies, reduces these pellets into metallic iron by removing oxygen from FeO to produce a product that is typically 92% to 96% metallized (Fe). Metallization is the percentage of metallic Fe out of the total iron content.
The rate of metallization depends on the DRI process and how it is operated, typically the higher the metallization rate, the lower the production rate and higher the specific consumptions rates for natural gas and electrical power. Further, in addition to gangue materials, the DRI will contain some quantity of carbon. In the EAF, this carbon can be used to first complete the reduction of the final fraction of the 4% to 8% iron oxide not reduced after which carbon in the product, can be useful for the formation of CO to produce foamy slag above the EAF’s melt line. Carbon is also of course needed to meet the required carbon content in the final steel produced, and it also, can be used to add chemical energy to the EAF to lower the electrical consumption of the EAF. Figure 3 indicates the composition of typical DRI produced at AM Montreal.
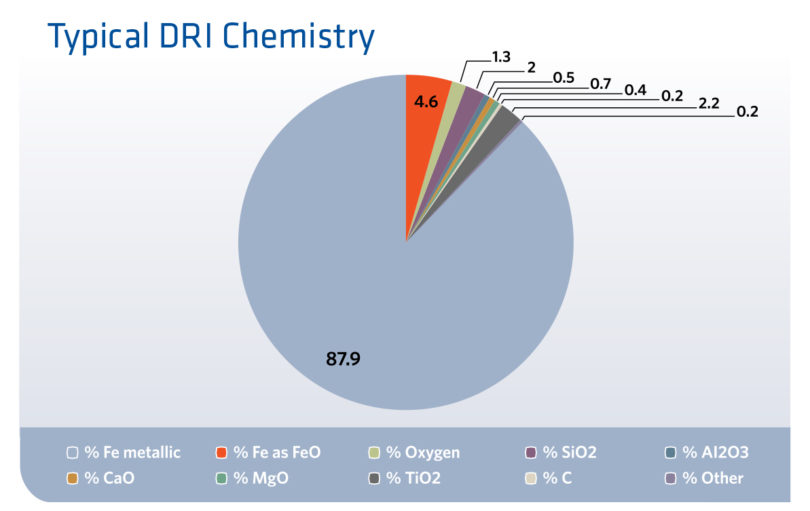
FIGURE 3.
Typical DRI Chemistry
One must look at the requirements of the meltshop and the economics of the DRI plant operation to determine what chemistry is optimum for his/her overall operation. Specifically the total iron content, degree of metallization and carbon content must be controlled to optimize the overall cost and throughput of the total cycle. Minimizing gangue content and reducing fines, (<5%) are beneficial regardless of the balance of the chemistry. Accordingly, for all practical purposes, the DRI quality can be defined based on two types of characteristics: those that are created by the DRI process such as degree of metallization, carbon content, and those that reside independent of the process such as the gangue content in the raw materials and fines concentration. See Figure 4*.

FIGURE 4.
What defines DRI Quality?
The relationship of changes in any one of these variables upon the EAF are indicated in Figure 5.
Ultimately the purpose of using DRI is to provide metallic iron to the meltshop, thus DRI is of better value to us if we can get more Fe to the meltshop. Metallization, fines, gangue and carbon all represent potential limitations to this goal.

FIGURE 5.
How does DRI Quality Influence Performance and Costs?
When addressing these properties, metallization, carbon and fines may be effected by the process. Whatever gangue, predominantly silica, is fed to the DRI plant is discharged as part of the product as the reduction process only removes oxygen from the product, not gangue materials. In any case, less is better when considering the total gangue in the DRI feed material. For reference see Figure 6 indicating the cost of increasing gangue content by one percent.
It should also be noted that fines can be created in the DRI process itself, but in this case we are referring more to the generation cause in normal handling of the raw materials and finished product. In any case, the more fines generated can be viewed as a loss of iron that could otherwise be utilized in the steelmaking process.
To minimize electricity consumption and increase efficiency, the goal is to have as much metallic iron as possible, meaning having a high rate of metallization (%Fe out of FeO). For every additional percent of metallized iron charged to an EAF there will be improvements in EAF yield, EAF productivity, EAF power consumption, refractory degradation and electrode degradation. Based on our experience at AM Montreal these relative effects are indicated in Figure 7.
As just stated, the real reason one uses or buys DRI is for its metallized iron content. Addition of any other component such as gangue materials and even carbon decreases the total iron in a charge. With that said, carbon is necessary in the EAF. The primary reasons are: reduction of the remaining non-metallized iron oxide in the DRI, formation of foamy slag, to meet the carbon specification in the steel being produced, and as additional chemical energy to the EAF thus reducing the electric power requirement. Figure 8 indicates the benefit of adding one full percent carbon to the EAF based on our experience at AM Montreal.
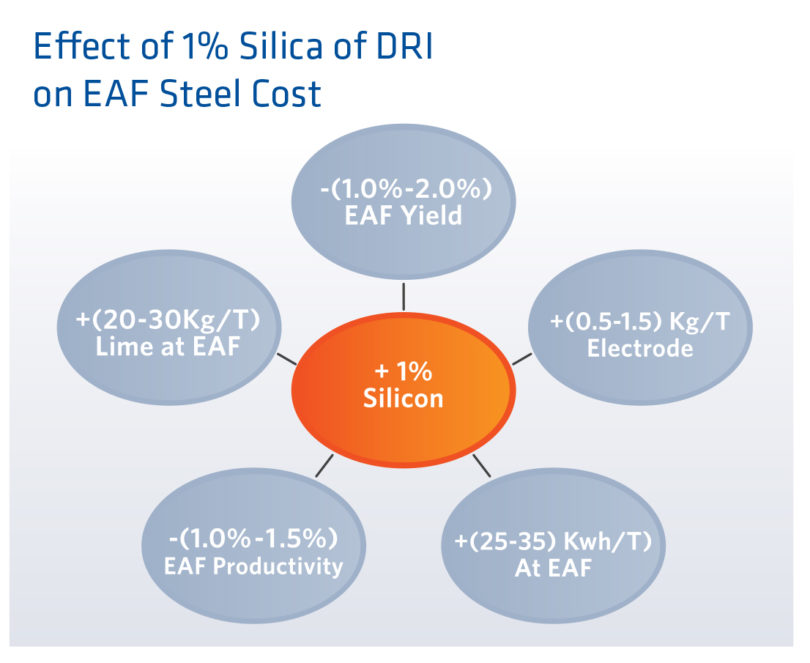
FIGURE 6.
Effect of Adding 1% Silica (Gangue) to DRI Feed Material
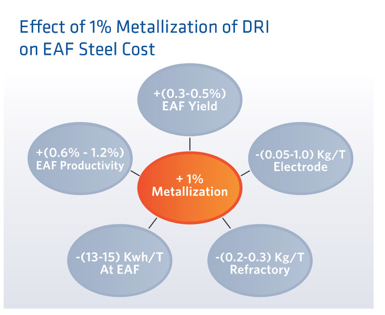
FIGURE 7.
Effect of Addition of 1% Metallization

FIGURE 8.
Effect of Addition of 1% Carbon
CONCLUSION
The value of the DRI is based on at least four major qualities, none of which are uniform, but all affect every operation. Our objective is to maximize liquid steel output from the EAF while reducing the utility costs to an optimum level. Over decades of operations we have determined that metallic content and physical quality are paramount. In any case we wish to start with the best raw materials (iron oxide feed materials) possible in order to have the least possible gangue and minimize fines generation. After that we need to maximize our Fe content even further through making DRI with high metallization. Based on our current utility, ore and scrap prices along with the production and quality demands of our onsite customer we are producing DRI at 94% to 95% metallization and 2.0% to 2.2% carbon. Ideally the EAF melt shop would prefer 95% to 96% metallization, but our DRI plants must do a continuous balancing act to assure optimum operations. Other AM DRI plants run at similar metallization rates with carbon levels ranging between 2.2% to 2.7% so even within our own company the optimum rates differ, yet the primary focus is still the Fe first. Please also note that in our scenario at AM Montreal we are looking at CDRI only, as that is reflective of our operations. HBI & HDRI share similar favorable qualities as CDRI, however, in the case of HDRI, temperature of the product is a major factor to increased EAF efficiency, without sacrificing metallic content.


